QC Batch Release Testing
Sciom delivers GMP-compliant QC batch release testing to ensure product safety, quality, and regulatory compliance before market release.
Request a QC Batch release Consultation
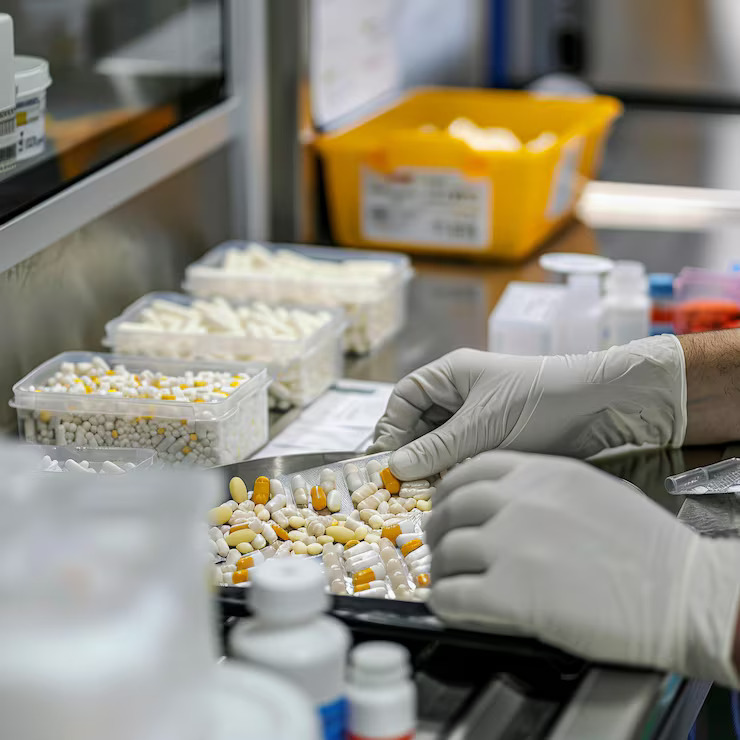
Ensuring every batch meets quality before release.
Our QC batch release testing ensures every batch is thoroughly evaluated for quality, safety, and compliance before release. From analytical testing to final approval, we help you meet regulatory expectations with confidence.
Every Test. Every Batch. Done Right.
Sciom delivers complete GMP QC testing for ALL dosage forms from one MHRA-approved UK laboratory – eliminating subcontracting delays and ensuring seamless compliance.
Oral Solids (Tablets/Capsules)
Assay, impurities, dissolution, disintegration, content/blend uniformity, hardness, friability, weight
variation
Liquids / Oral Solutions
Assay, impurities, pH, viscosity, density, dissolution, appearance (diffused daylight cabinet),
preservative efficacy, antimicrobial effectiveness
Injectables / Steriles
Endotoxins (LAL), sub-visible particles, visible particles, osmolality
Raw Materials / API / Excipients
Identification, assay, residual solvents, heavy metals, water (Karl Fischer), LOD, related substances
Microbiological Testing
Microbial enumeration, specified organisms, endotoxins, environmental monitoring, preservative efficacy
testing
Instrumental Analysis
HPLC / GC chromatography, UV/IR spectroscopy, LC-MS, GC-MS, detectors (DAD, VWD, PDA, FID, RID)
Every step tracked. Every result trusted.
Secure sample intake with full chain-of-custody documentation under controlled conditions. Methods (Ph. Eur./BP or client-specific) are verified, and samples are logged in Pharma Plus with unique IDs for complete traceability via client portal.
A Standard Testing Procedure (STP) is generated outlining tests, methods, and specifications. Each STP is barcoded to ensure accurate tracking and data integrity.
GMP-compliant testing is performed using validated instruments, covering chemical, physical, and microbiological analysis. Data is captured in real-time for accuracy and audit readiness.
Results undergo QA review, including raw data and audit trails. Any OOS results are investigated, and only compliant data is approved.
A CoA is generated and approved, presenting results against specifications. Clients can access reports via a secure portal for quick decisions.
All records are stored in a 21 CFR Part 11-compliant system with full audit trails, ensuring ALCOA+ data integrity and long-term compliance.
Compliance at every step. No exceptions.

Sciom ensures regulatory-compliant QC batch release testing, where every batch is evaluated against the approved Marketing Authorisation, with full transparency, traceability, and alignment to global standards.
Regulatory Compliance Capabilities:
Built for precision. Backed by expertise.
State-of-the-art analytical platforms including HPLC, GC, LC-MS, GC-MS, UV-Vis, FTIR, and dissolution systems enable comprehensive QC batch release testing. Dedicated chemical and microbiological laboratories support accurate, high-quality analysis, while scalable facilities handle both routine and complex testing programs.
End-to-end support for analytical method development, validation, and transfer, ensuring methods are fit-for-purpose and regulatory compliant. All processes align with ICH guidelines and global regulatory standards, enabling reliable testing for diverse product types.
Comprehensive chemical and physical testing services, including assay, related substances, residual solvents, dissolution, and material characterization. Advanced impurity profiling ensures product quality, consistency, and compliance with regulatory specifications.
Robust microbiological testing including microbial limits, bacterial endotoxins, and preservative efficacy testing. Controlled environments and environmental monitoring systems ensure compliance with GMP and aseptic processing requirements.
Trusted expertise for reliable batch release
Replace multiple providers for QC, microbiology, nitrosamine testing, importation, and QP release with one seamless service—saving time, effort, and complexity.
Enjoy secure, dedicated portal access for full traceability of your batch status—from sample receipt to QP release. Track progress in real-time with ETA for every sample.
No more follow-up emails or phone calls—you can track progress in real-time with ETA for every sample.
Deviations, CAPAs, change controls, complaints, and training are managed electronically in a centralized system, ensuring efficiency, accuracy, and compliance.
Our eQMS reduces documentation turnaround by 60% compared to traditional paper-based processes, helping your batches move faster without compromising quality.
Everything you need. One trusted partner.
Stay informed with expert commentary on pharmacovigilance services, regulatory consulting services, analytical science and audit compliance services across the pharmaceutical industry.